 
In a molecule of O2, there are two sigma bonds and two pi bonds. 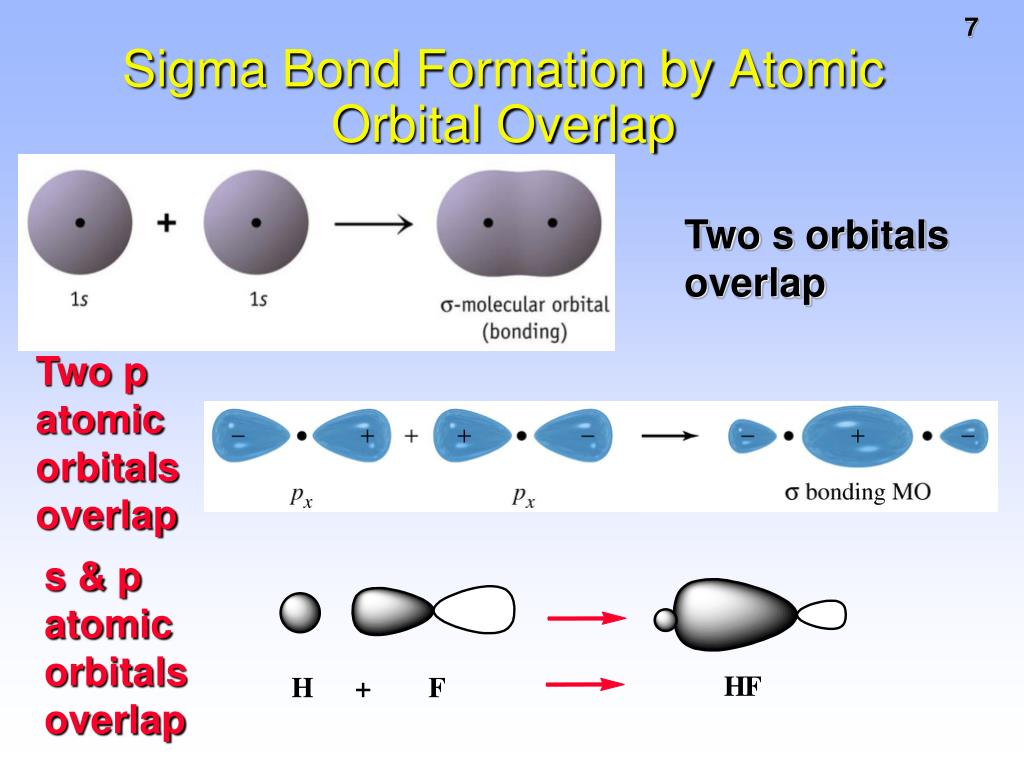
The two atoms share one pair of electrons in a sigma bond. In a molecule of H2, there is only one sigma bond. Now that we know what sigma and pi bonds are, let's take a closer look at some examples in different molecules. Three types of sigma bonds between s-s, s-p, and p-p atomic orbitals and a pi bond between p-p orbitals Sigma bonds are stronger and can exist independently in single bonds, while pi bonds must coexist with a sigma bond and are only found in double and triple bonds. In summary, sigma and pi bonds are types of covalent bonds formed by different types of atomic orbital overlap. When two molecules bond, their orbitals usually combine to form hybrid orbitals like sp, sp2, and sp3. There are four types of atomic orbital sets: s, p, d, and f. Atomic orbitals are spaces where electrons are likely to be found. To understand sigma and pi bonds, you need to know a little about atomic orbitals and hybridization. Second, sigma bonds can exist independently in single bonds, while pi bonds must coexist with a sigma bond and are only found in double and triple bonds. So, what's the difference between sigma and pi bonds? First, sigma bonds are stronger than pi bonds. They only exist in double and triple bonds. Pi bonds (π) are the second and third types of covalent bonds, formed by overlap of p orbitals side-to-side. They are found in single, double, and triple bonds. Sigma bonds (σ) are the first type of covalent bond, formed by overlap of atomic orbitals head-to-head. Covalent bonds happen when atoms share electrons. But actually, sigma and pi bonds are types of covalent bonds. Of course, there exist #f# orbitals of some sort that can overlap with #s#, #p_z#, #d_(z^2)#, and #d_(x^2-y^2)# in a #sigma# fashion, but that's left up to the really motivated chemist to figure out.When you hear the words sigma and pi bond, you might think of Greek life in college. (Since #s# orbitals are spheres, it doesn't matter along which axis they bond.) Since we supposed that only the #sigma# bonding MO is occupied, we have a single bond. Since they are all aligned along the same axis ( #p_z# with #d_(z^2)#, #p_x# with #d_(x^2-y^2)#, and #p_y# with #d_(x^2-y^2)#) AND they are compatible ( #s# with #p_z#, #s# with #d_(z^2)#, and #s# with #d_(x^2-y^2)#), they form #sigma# bonding and #sigma^"*"# antibonding MOs. Therefore, they overlap with the #p_x# and #p_y#, which also lie long those axes. The opposite lobes along each axis are the same sign. The #d_(x^2-y^2)# can be approximated as four-leaf clovers, essentially, on the #xy#-plane, with the lobes aligned along the #x# and #y# axes.You can also approximate these as dumbbells, regardless of their #n#, without losing the essence of the #sigma# MOs generated (head-on overlap). The #d_(z^2)# look almost like #p_z# orbitals, except there is a donut in the middle.One lobe is the opposite sign to the other. The #p_z# orbitals can be approximated as dumbbells, regardless of their #n#, without losing the essence of the #sigma# MOs generated (head-on overlap).The only way these can change sign is if the whole thing changes sign. HOW TO DEPICT/IMAGINE THESE ORBITAL OVERLAPS We would also suppose that the antibonding molecular orbitals are unoccupied so that the bond is a standard single bond. the axis along which the single bond-which is also a #sigma# bond-is made), and the #x# and #y# axes are where you should expect them to be for the Cartesian coordinate system. Where the #z# axis is the internuclear axis (i.e. Then, orbitals that can capably overlap with each other to form #sigma# bonds include the following linear combinations: Let's also suppose that we are ignoring #d#- #d# interactions, since we know those should work (they are the same #l# so it's not so interesting). POSSIBLE ORBITAL COMBINATIONS TO GENERATE SIGMA MOLECULAR ORBITALSįor simplicity, if we examine only the #s#, #p#, and #d# orbitals, let's suppose that all orbitals we are examining are similar enough in energy to interact. #sigma# bonds must be made by orbitals that overlap head-on. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
